The FDA-approved drug Auranofin has a dual inhibitory effect on SARS-CoV-2 entry and NF-ÎşB signaling
iScience, Volume 25, Issue 10, 2022, 105066.
Emmanuel Laplantine, Christine Chable-Bessia, Anne Oudin, Jitendryia Swain, Adèle Soria, Peggy Merida, Manon Gourdelier, Sarra Mestiri, Indira Besseghe, Erwan Bremaud, Aymeric Neyret, Sebastien Lyonnais, Cyril Favard, Philippe Benaroch, Mathieu Hubert, Olivier Schwartz, Maryse Guerin, Anne Danckaert, Elaine Del Nery, Delphine Muriaux, Robert Weil.
ISSN 2589-0042,
https://doi.org/10.1016/j.isci.2022.105066.
(https://www.sciencedirect.com/science/article/pii/S2589004222013384)
Abstract:
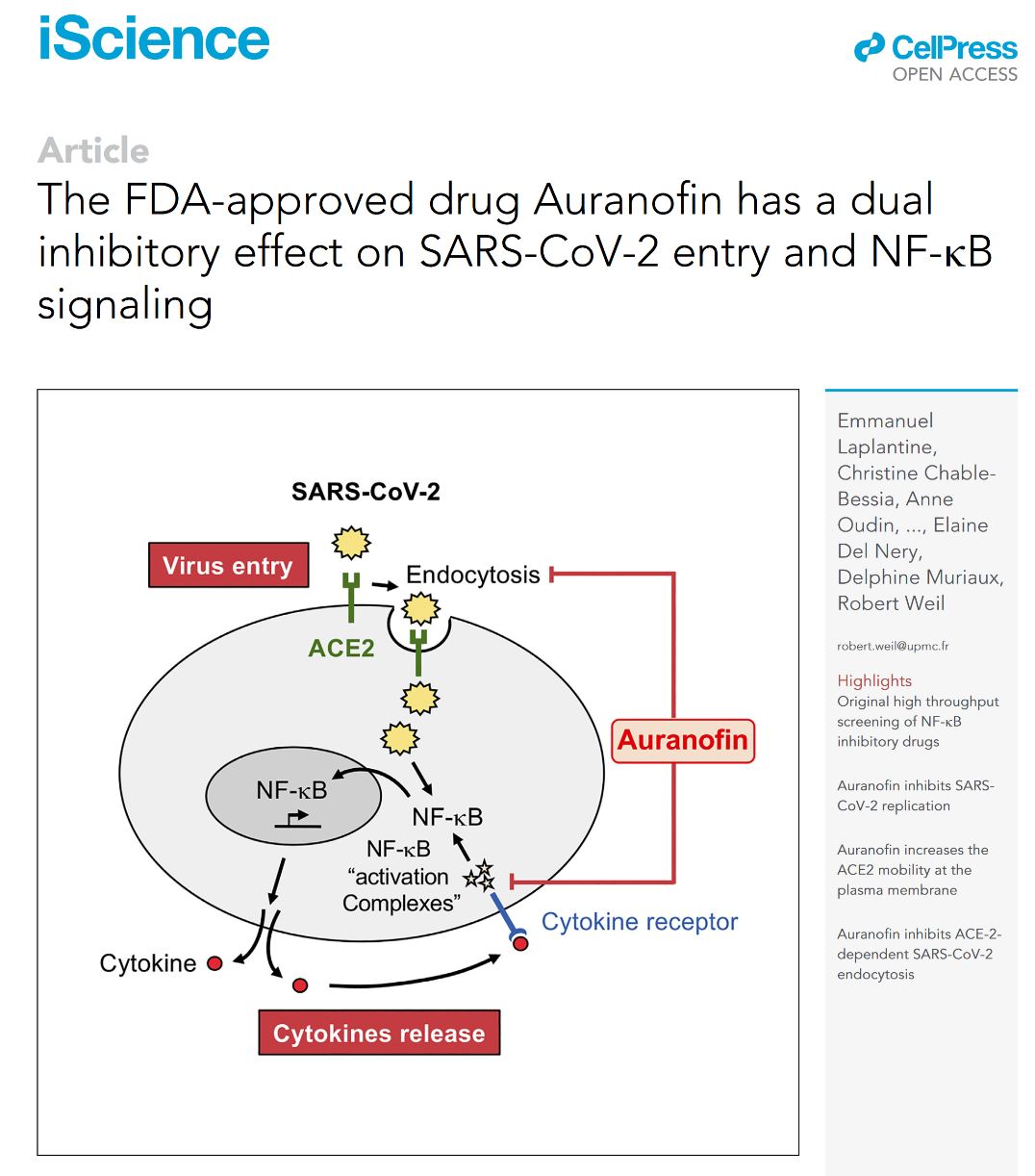
Patients with severe COVID-19 show an altered immune response that fails to control the viral spread and suffer from exacerbated inflammatory response, which eventually can lead to death. A major challenge is to develop an effective treatment for COVID-19. NF-ÎşB is a major player in innate immunity and inflammatory process. By a high-throughput screening approach, we identified FDA-approved compounds that inhibit the NF-ÎşB pathway and thus dampen inflammation. Among these, we show that Auranofin prevents post-translational modifications of NF-ÎşB effectors and their recruitment into activating complexes in response to SARS-CoV-2 infection or cytokine stimulation. In addition, we demonstrate that Auranofin counteracts several steps of SARS-CoV-2 infection. First, it inhibits a raft-dependent endocytic pathway involved in SARS-CoV-2 entry into host cells; Second, Auranofin alters the ACE2 mobility at the plasma membrane. Overall, Auranofin should prevent SARS-CoV-2 infection and inflammatory damages, offering new opportunities as a repurposable drug candidate to treat COVID-19.



 Chargement en cours ...
Chargement en cours ...